The electron configuration for the first 10 elements. So, a number of valence electrons in lead is 4. Electron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table. The concept of electronic configuration has replaced the older concept of valency and valence electrons. But the valency of elements, when combined with H or O first, increases from 1 to 4 and then it reduces to zero. The electronic configuration of each element is decided by the Aufbau principle which states that the electrons fill orbitals in order of increasing energy levels.
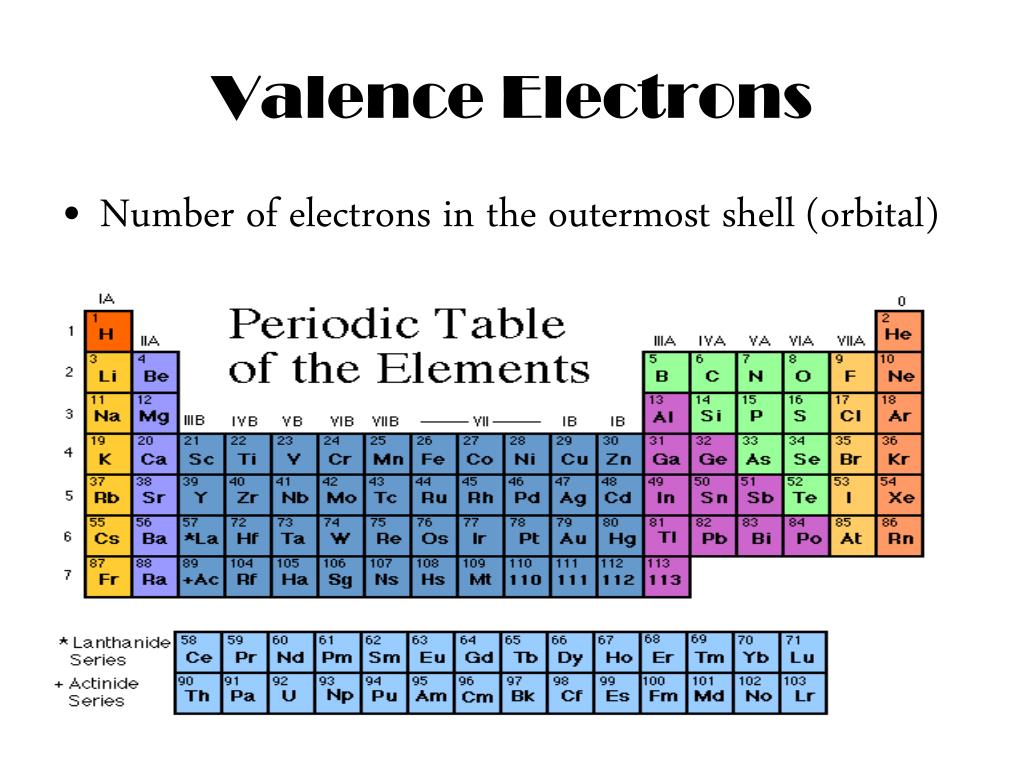
Elements in other groups have partially filled valence shells and gain or lose electrons to achieve a stable. A full valence shell is the most stable electron configuration. Group 18 elements (helium, neon, and argon are shown in Figure 2) have a full outer, or valence, shell. The electron configurations of the elements indicated in blue are also anomalous, but the reasons for the observed configurations are more complex. While moving left to right across a period, the number of valence electrons of elements increases and varies between 1 to 8. Bohr diagrams indicate how many electrons fill each principal shell. Variation Of Oxidation State Along a Period. In case of lead, there are 4 electrons present in the 6 th or outer shell. Periodic Trends in the Oxidation States of Elements. How Many Valence Electrons Does Lead haveĪ number of valence electrons are defined as a number of electrons present in the outer shell of atom or molecule. And electron configuration of Pb (lead) is defined as 1s 22s 22p 63s 23p 63d 104s 24p 64d 104f 145s 25p 65d 106s 1.

What is The Electron Configuration of LeadĮlectrons distribution in the orbit of atom or molecule is termed as electron configuration. Valence electrons are electrons in the outer most shell or orbit and there are 4 valence electrons present in the lead (Pb) outermost shell.įull electron configuration for lead is 1s 22s 22p 63s 23p 63d 104s 24p 64d 104f 145s 25p 65d 106s 1. The noble gas configuration encompases the energy states lower than the valence shell electrons. The periodic table shows that kyrpton (Kr) is the previous noble gas listed before iodine. 4f 145d 106s 26p 2 Lead Valence Electrons In the short notation, you place brackets around the preceding noble gas element followed by the valence shell electron configuration.
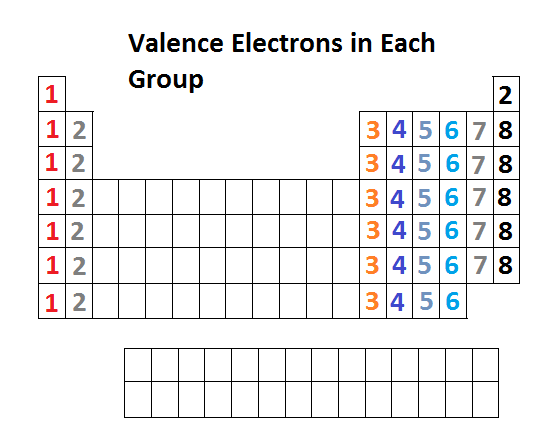
The distribution of electrons is 2, 8, 18, 32, 18, and 4.Įlectronic configuration for lead can be written as: 1s 22s 22p 63s 23p 63d 104s 24p 64d 104f 145s 25p 65d 106s 26p 2 This element has 82 number of electrons present in its 6 orbits.
